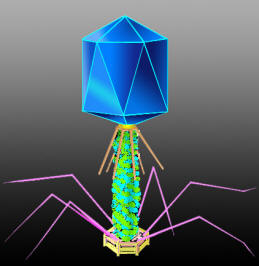
Vinco-T4
Phage size does Matter…
It represents the maximum potential capacity of the insertion length (bp). Small phage particle cannot afford the over-size DNA cloning. 

publishcation
1). Ren, Z.J., et al. (1996): Phage display of intact domains at high copy number. A system based on SOC, the small outer capsid protein of bacteriophage T4. Protein Science, 5: 1833-1843.
2). Ren, Z.J., et al. (1997) Cloning of linear DNAs in vivo by overexposed T4 DNA ligase: construction of a T4 phage hoc gene display vector. Gene, 195: 303-311.
3). Ren, Z.J., and Black, L.W (1998): Phage T4 SOC and HOC display of biologically active, full-length proteins on the viral capsid. Gene, 215 :439.
4). Wu,J.M.,et. al..(2004): Bacteriophage T4 capsid surface Soc and Hoc sites bipartite display: CSFV immunogenicity comparison with plasmid expressed antigen. (submitted to Infection and Immunity)
5). Ren,Z.J, et al.: Development of a novel FMDV oral vaccine: T4 phage-engineering with full-length capsid gene P1 and proteinase gene 3C both expressed on viron surfice. (Vaccine, in preparation )
6). Cao,y.C.,et al..: A vaccine condidate: Display of viral structural protein VP2 and HV of infectious bursal disease virus( IBDV) on the Soc site of phage T4 capsid surface. ( Vaccine, in preparation )
7). Ren, Z.J.,et al. : Capture of food-and-mouth disease virus antigenic domains and expression on T4 phage viron surface. ( Virology, in preparation )
8). Zhao,M.Y.,et al.: T4 -FMDV phage survival virons distribution, processing in mice organs after oral administration ( J. Virology, in preparation )
9). 吴健敏、任兆钧、余兴龙等. 中国兽医学报, 2003, 23 (1):14-17
10). Jiang, J., et al.. (1997): Display of a PorA peptide from Neisseria meningitidis on the bacteriophage T4 capsid surface, Infec. and Imm. 65:4770-4777
11). Naglis Malys, et al. (2002), A bipartite bqacteriophage T4 SOC and HOC randomized peptide display library: detection and analysis of phage T4 terminase (gp17) and late ơ factor (gp55) interaction, J.Mol.Biol. 319: 289-304.
12). Malmborg AC (1999) Phage display method. In: Real-time analysis of biomolecular interactions - applications of BIACORE (English edition). (Eds.: Nagata K, Handa H ) Springer-Verlag, Tokyo. pp. 187-194.
13). Yanagida,M.(1997): Molecular organization on the shell of T even bacteriophage head. II. Arrangement of subunits in the head shells of giant phages. J. Mol. Biol. 109: 513-537.
14). McCafferty J, (1990): Phage antibodies: filamentous phage displaying antibody variable domains. Nature ,348:552-554.
15). Forrer P, Jaussi R (1998): High-level expression of soluble heterologous proteins in the cytoplasm of Escherichia coli by fusion to the bacteriophage lambda head protein D.Gene , 224: 45-52.
16). Daugherty PS, et al. (1998): Antibody affinity maturation using bacterial surface display. Protein Eng, 11:825-832.
17). Kieke MC,(1997): Isolation of anti-T cell receptor scFv mutants by yeast surface display. Protein Eng, 10:1303-1310.
18). Hanes J, Pluckthün A (1997): In vitro selection and evolution of functional proteins by using ribosome display.Proc Natl Acad Sci USA, 94:4937-4942.
19). Marks JD, et al. (1991) : By-passing immunization. Human antibodies from V-gene libraries displayed on phage. J Mol Biol, 222:581-597.
20).Vaughan TJ, (1996): Human antibodies with sub-nanomolar affinities isolated from a large non-immunized phage display library. Nat Biotechnol, 14:309-314.
21). Sheets MD, et al (1998): Efficient construction of a large nonimmune phage antibody library: the production of high-affinity human single-chain antibodies to protein antigens.Proc Natl Acad Sci USA, 95:6157-6162.
22). De Haard HJ,et al. (1999): A large non-immunized human Fab fragment phage library that permits rapid isolation and kinetic analysis of high affinity antibodies. J Biol Chem, 274:18218-18230.
23). Little M, (1999) Generation of a large complex antibody library from multiple donors.J Immunol Methods, 231:3-9.
24). Hoogenboom HR, Winter G (1992): By-passing immunisation. Human antibodies from synthetic repertoires of germline VH gene segments rearranged in vitro. J Mol Biol, 227:381-388.
25). Akamatsu Y, et al. (1993): Construction of a human Ig combinatorial library from genomic V segments and synthetic CDR3 fragments. J Immunol, 151:4651-4659.
26). Nissim A, et al (1994): Antibody fragments from a ‘single pot’ phage display library as immunochemical reagents.EMBO J , 13:692-698.
27). De Kruif J, Boel E, Logtenberg T (1995): Selection and application of human single chain Fv antibody fragments from a semi-synthetic phage antibody display library with designed CDR3 regions. J Mol Biol, 248:97-105.
28). Knappik A, et al (2000) : Fully synthetic human combinatorial antibody libraries (HuCAL) based on modular consensus frameworks and CDRs randomized with trinucleotides. J Mol Biol, 296:57-86.
29). Pini A, et al (1998): Design and use of a phage display library. Human antibodies with subnanomolar affinity against a marker of angiogenesis eluted from a two-dimensional gel. J Biol Chem, 273:21769-21776.
30). Lucy J Holt, et al. (2000): The use of recombinant antibodies in proteomics. Current Opinion in Biotechnology 2000, 11:445–449
31). Sheets, M. D., et al. (1998): Efficient construction of a large nonimmune phage antibody library: the production of high-affinity human single-chain antibodies to protein antigens. Proc. Natl. Acad. Sci. USA, 95, 6157-6162.
32). De Haard HJ, et al (1999): A large non-immunized human Fab fragment phage library that permits rapid isolation and kinetic analysis of high affinity antibodies. Proc. Natl. Acad. Sci. USA, 96(2): 795